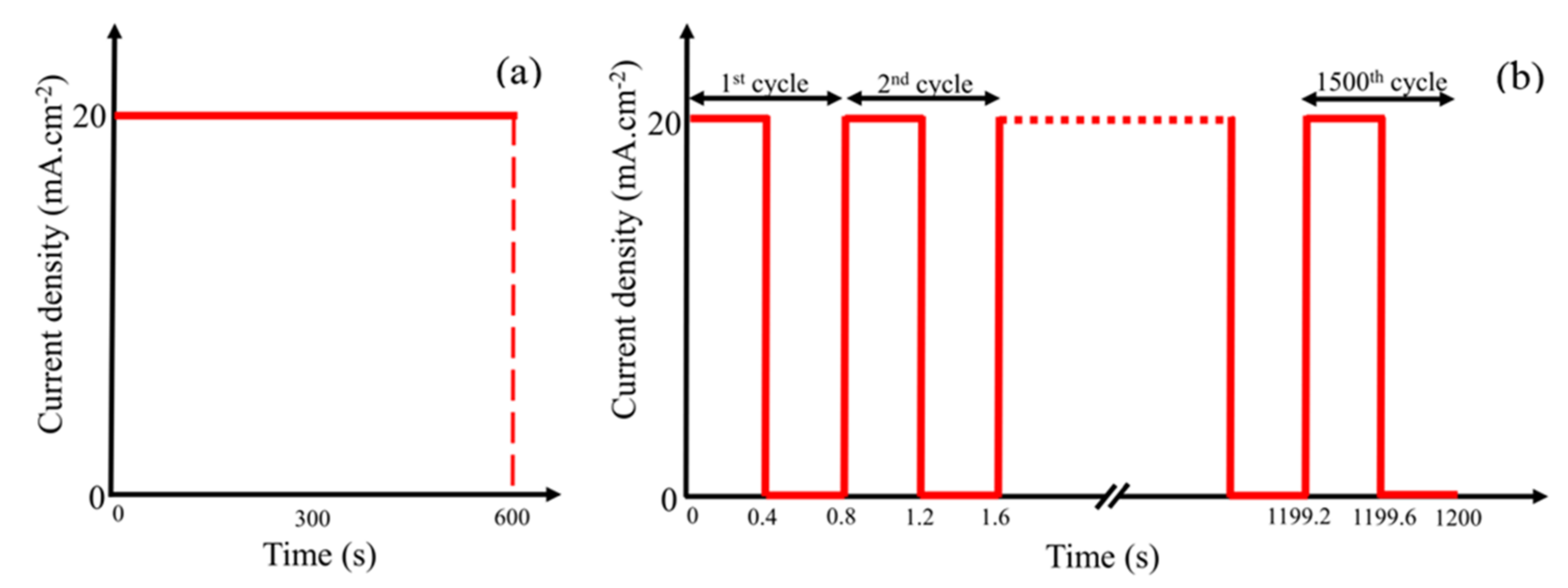
1–4 The demands on higher energy density stimulate the development of LIBs.
ACCELRYS MATERIALS STUDIO 4.3 PORTABLE
Introduction Rapid progress in the energy industry leads to the wide application of lithium-ion batteries (LIBs) as portable or stationary energy storage devices due to their advantages in terms of energy density, energy efficiency and moderate working temperature.
The comprehensive understanding of the solution structure and properties is necessary for the rational design of advanced electrolytes.
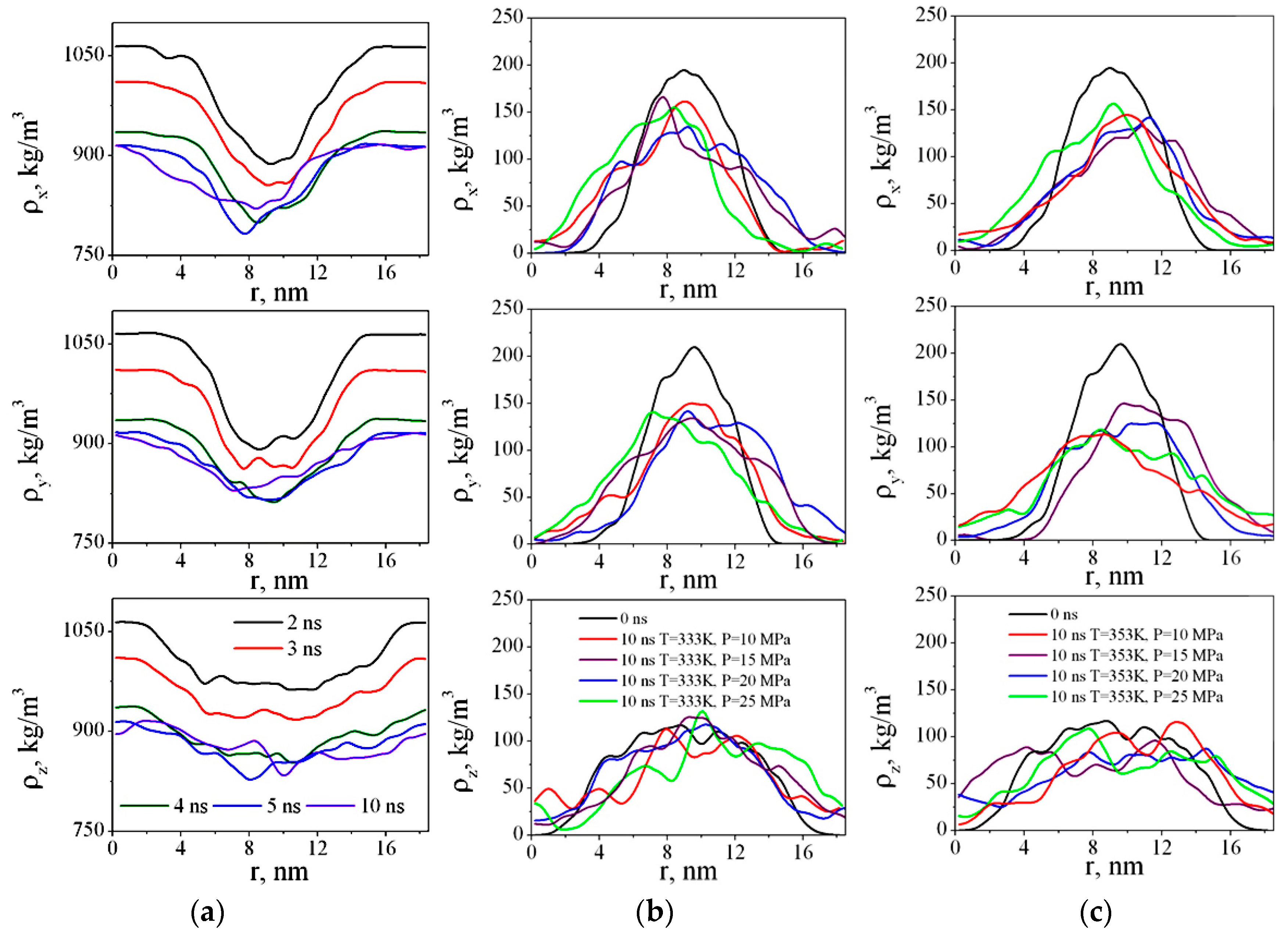
The results indicate that the electronic structure of anions contributes greatly to the charge distribution of the Li + solvation sheath, and consequently extends to the thermodynamics of the carbonate molecules, affecting reduction, oxidation reaction and products on the interface between electrolytes and electrodes. With different anions, the electrolytes are different in possible solvation structures and charge distributions, leading to differences in terms of thermal properties, viscosity, ionic conductivity, electrochemical oxidation and reduction behaviors as well as LiNi 0.6Mn 0.2Co 0.2|Li cell performances. The anion effect on Li + solvation structure and consequent electrochemical and physical properties was studied on the basis of LiFSI-DMC (lithium bisfluorosulfonyl imide-dimethyl carbonate)- and LiTFSI-DMC (lithium bis(trifluoromethanesulfonyl imide)-dimethyl carbonate)-based dilute electrolytes, highly concentrated electrolytes, and localized concentrated electrolytes.
